The pharmaceutical industry faces unique challenges in trademark clearance, including complex regulatory requirements and the need for rapid, precise name approvals. To address these needs, Corsearch offers innovative tools that streamline and optimize the trademark clearance process for pharma professionals.
In this blog, we explore how Corsearch’s PharmaCheck™ approach and TrademarkNow solutions can help pharmaceutical companies save time, reduce costs, and navigate the evolving trademark landscape with confidence.
Changing landscape
The Trademark Modernization Act (TMA) of 2020 set the scene for trademark registrants and applications to progress application, registration, and litigation far more quickly – and by simpler and cheaper means than were available previously.
The three key changes introduced were to the options and process for ex parte proceedings, prosecution, and litigation. The USPTO established new protocols and procedures that allow third parties to ease some registration backlogs by having marks expunged or re-examined, at reasonable cost, and in addition to similar action taken by the USPTO of its own volition – particularly when such marks are not in current commercial use. Those with actions to bring on the litigation and prosecution sides should find the USPTO processes streamlined and easier to work with.
Although the changes make trademark challenges easier and more cost-effective to initiate, there remain sufficient protections in place for genuine and active registrants, and there are double jeopardy rules to ensure that a registrant should not find themselves serially forced to prove use.
Tools for the job
Corsearch’s upgraded portfolio of trademark clearance support tools has been developed with the streamlining and expedience of the new legislative and regulatory framework in mind. What was previously a labor-intensive and potentially costly exercise should now be a faster, cheaper, and more efficient end-to-end process.
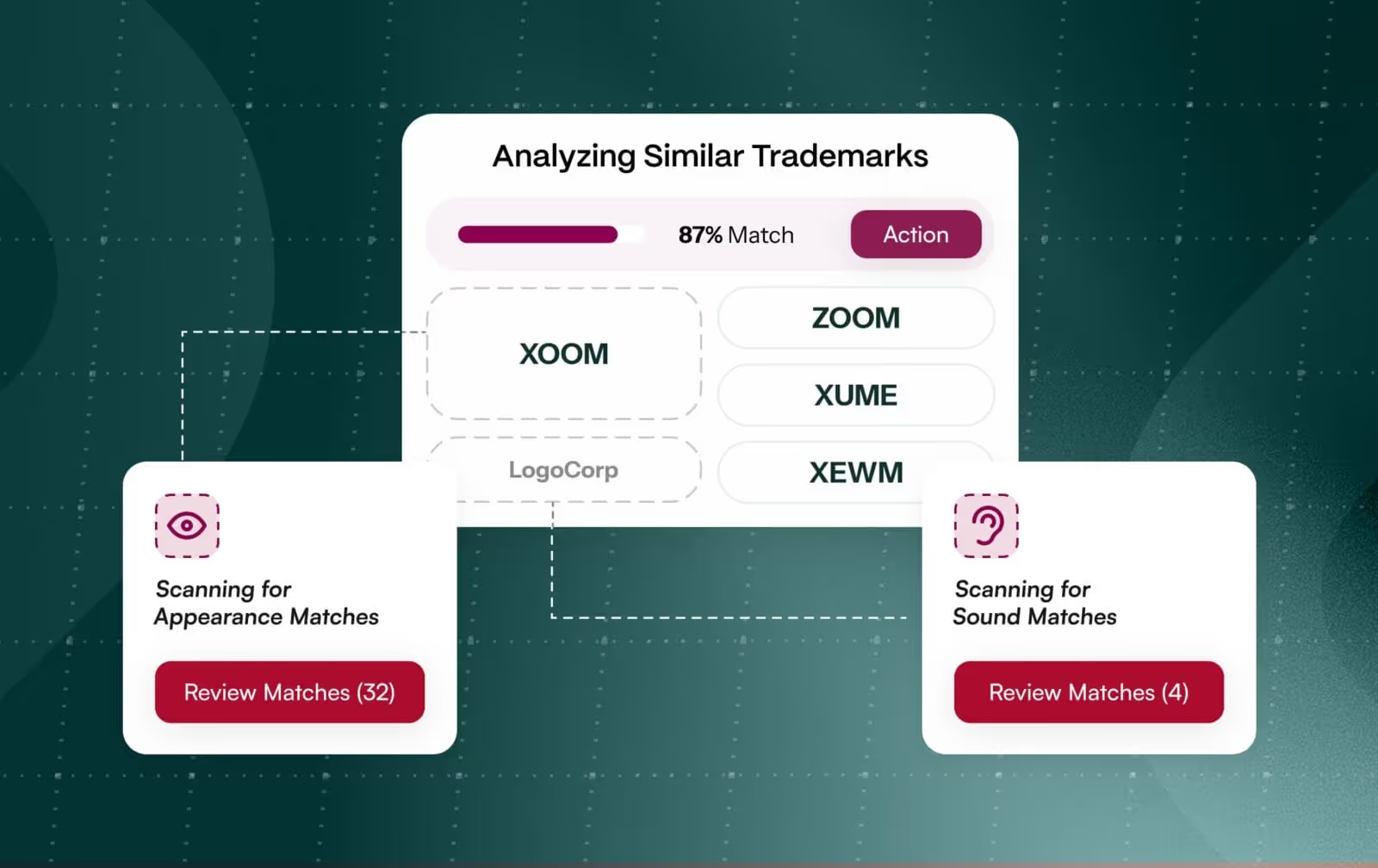
Our PharmaCheck™ approach combines complementary tools and access to relevant databases. Using the FDA’s Phonetic and Orthographic Computer Analysis (POCA) algorithm speeds up the search for pharma and regulatory sources, with simultaneous access to over 40 databases of registry, pharma in-use, regulatory, domain names, and USAN/INN stems (at risk of piggybacking) ensures all avenues are explored during the clearance discovery process. We can cross-compare entries in different databases, and process up to 150 candidate names in a single bulk screening search.
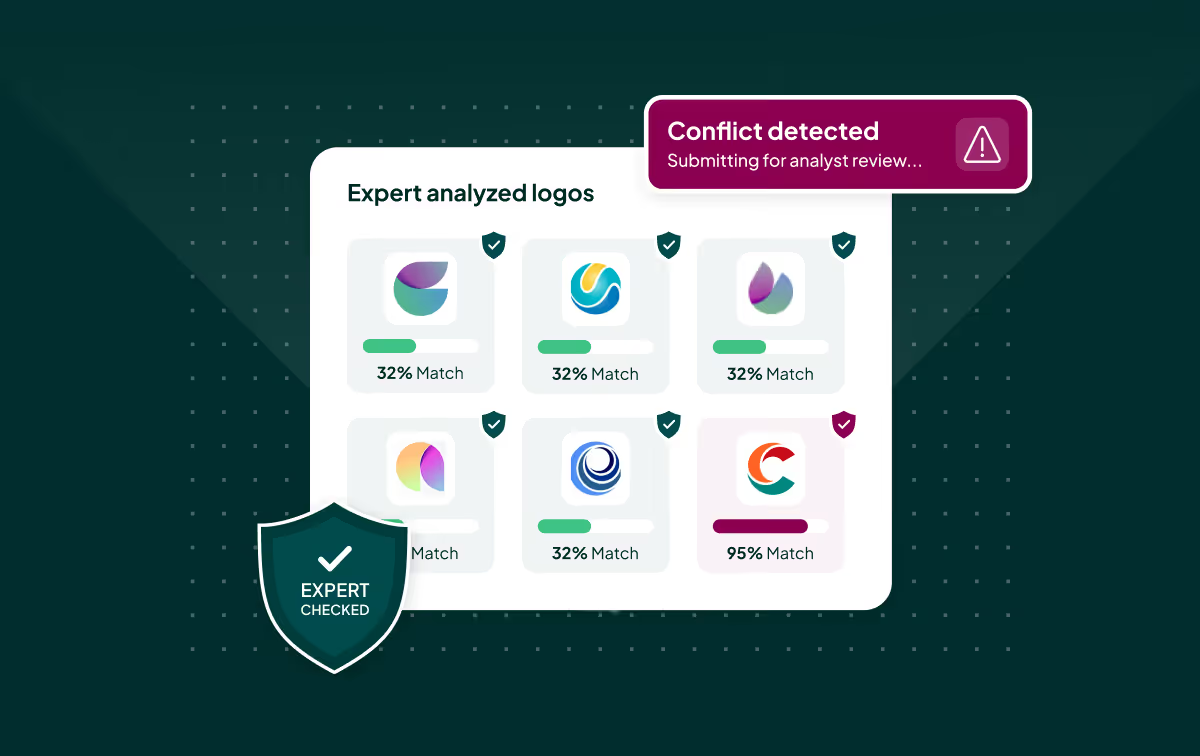
Our TrademarkNow solution provides access to over 185 global trademark databases, and comprises four powerful AI tools: Examatch, for instant knockout and business intelligence; Portfolio Analyzer, to compare entire portfolios against the competition and industry benchmarks; LogoCheck, utilizing the latest image-recognition technology; and NameCheck, to iron out similarity issues on the final path to clearance.
Finally, our expert Watch solutions help registrants maintain the unique value of their marks, by monitoring new global trademark filings and publications of marks that may be confusingly similar to yours, and helping to trigger decisions to oppose or take action.
Talk to an expert
If you would like to explore how our pharma toolset can save your team time and money, please talk to one of our experts.
See how PharmaCheck™ accelerates name qualification
From early-stage screening to post-clearance checks, we help you move faster with confidence — while avoiding missteps new, and help you move forward — faster.








.svg)
.avif)








.svg)