Confident Name Screening for Regulated Pharma Environments
Our platform brings together trademark and regulatory clearance into a single, intuitive workflow, helping you and your brand to:

Accelerate Viable Name Selection
By screening trademarks and regulatory data (Pharma-in-Use, INN/USAN, POCA search results, marketing authorizations from FDA, EMA, SwissMedic, Health Canada, and more) in one place.

Improve Cross-functional Alignment
Worldwide reach with local insights – proven success across platforms to unify your workflows.Worldwide reach with local insights – proven success across platforms to unify your workflows.

Minimise Launch Risk
By mirroring regulatory authority logic (POCA scoring) and identifying lookalike/ soundalike conflicts early.
180+
11
75%
15+
From First Screening to Final Decision
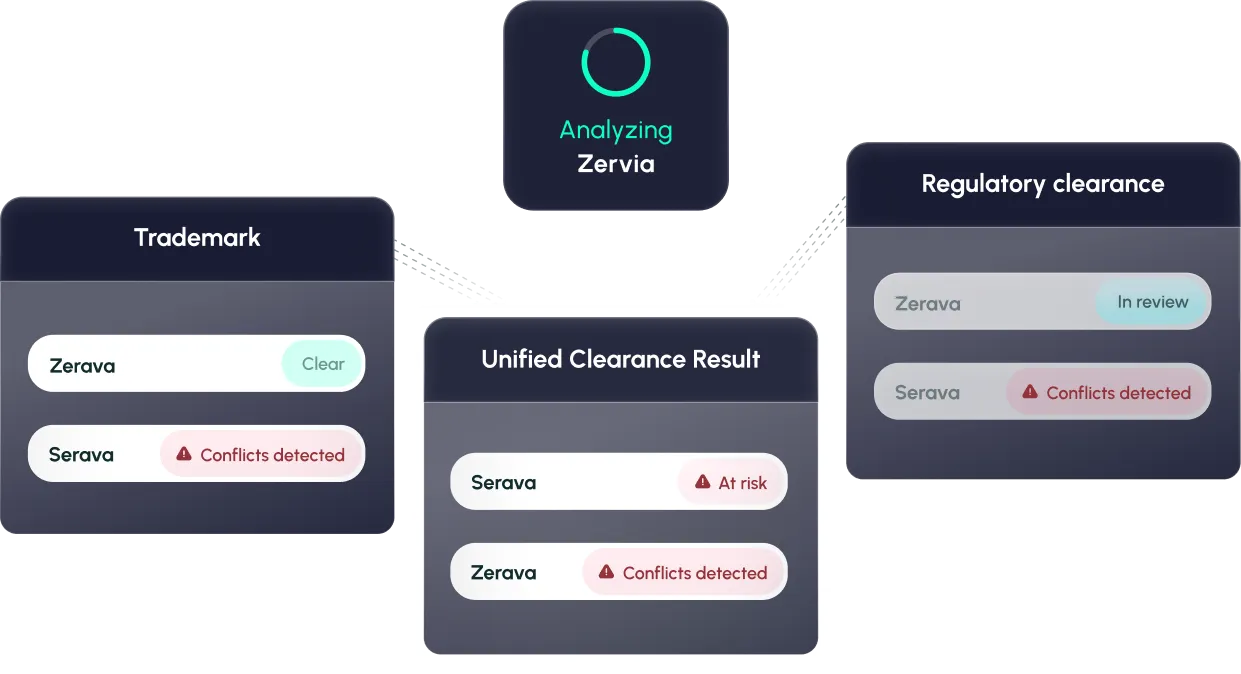
Unified Clearance
Search across global trademarks, pharma-specific sources (PIU, INN/USAN, marketing authorizations), and get results scored in real time.
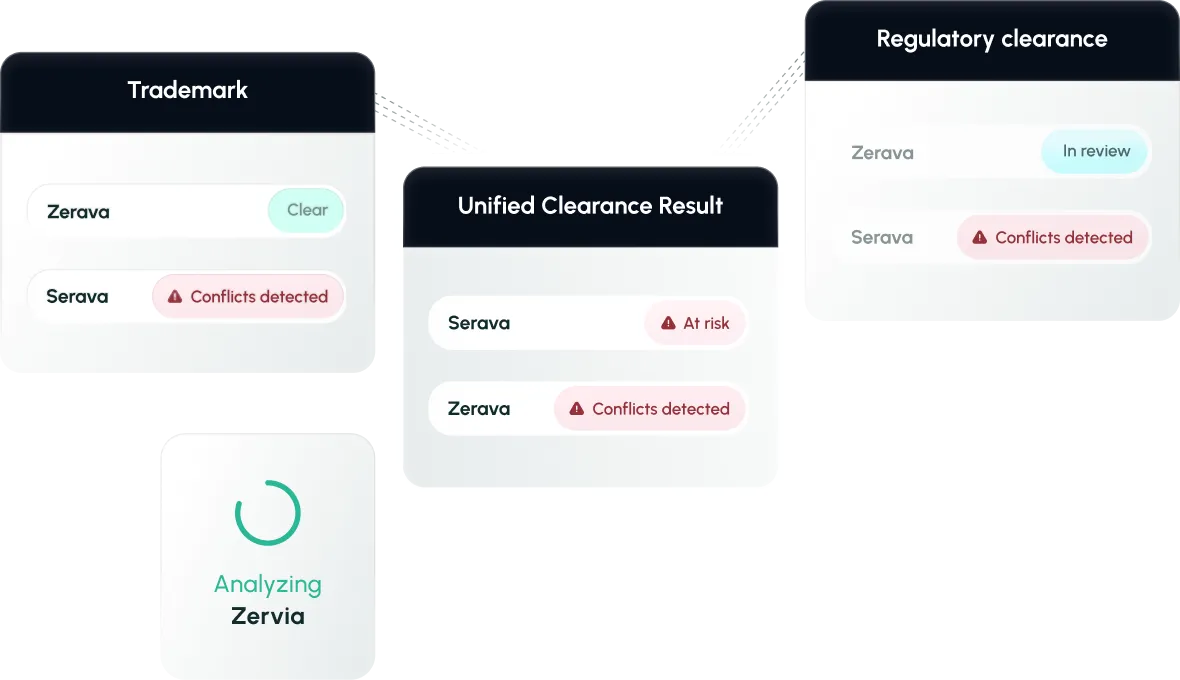
Smarter Risk Scoring
Compare candidate names by combined POCA, phonetic, or orthographic scores - aligned with regulatory expectations.
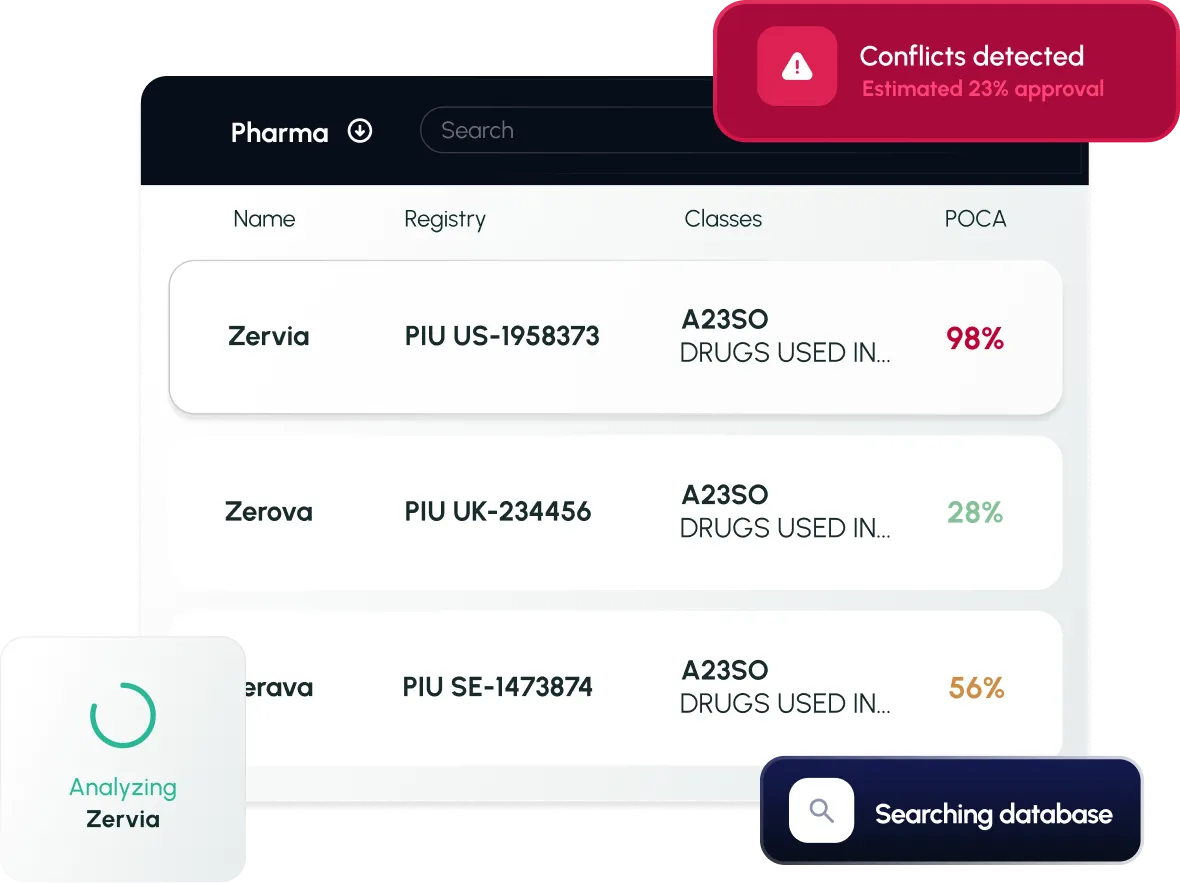
Workflow Continuity
Build on prior searches, connect results, export reports, and access product-level insights like sales revenue, launch timelines, and ownership.
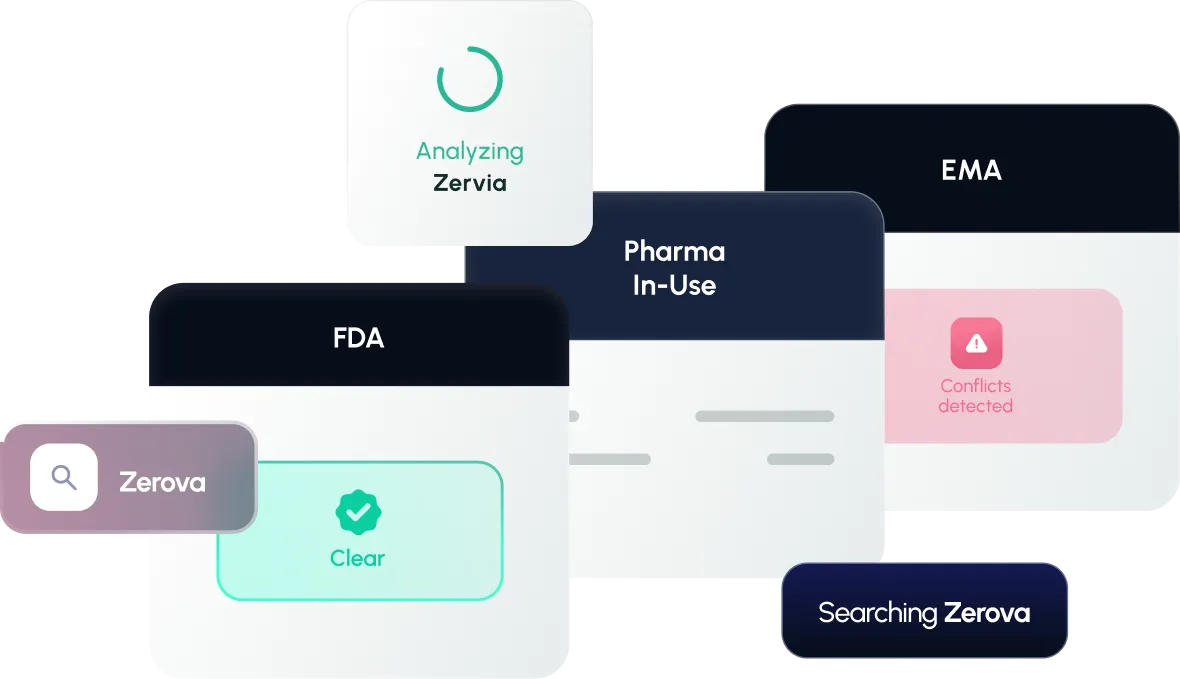
Trademark Solutions across the Lifecycle
We meet you where you are, and deliver the right pharma marks solution for every phase.









.svg)
.avif)